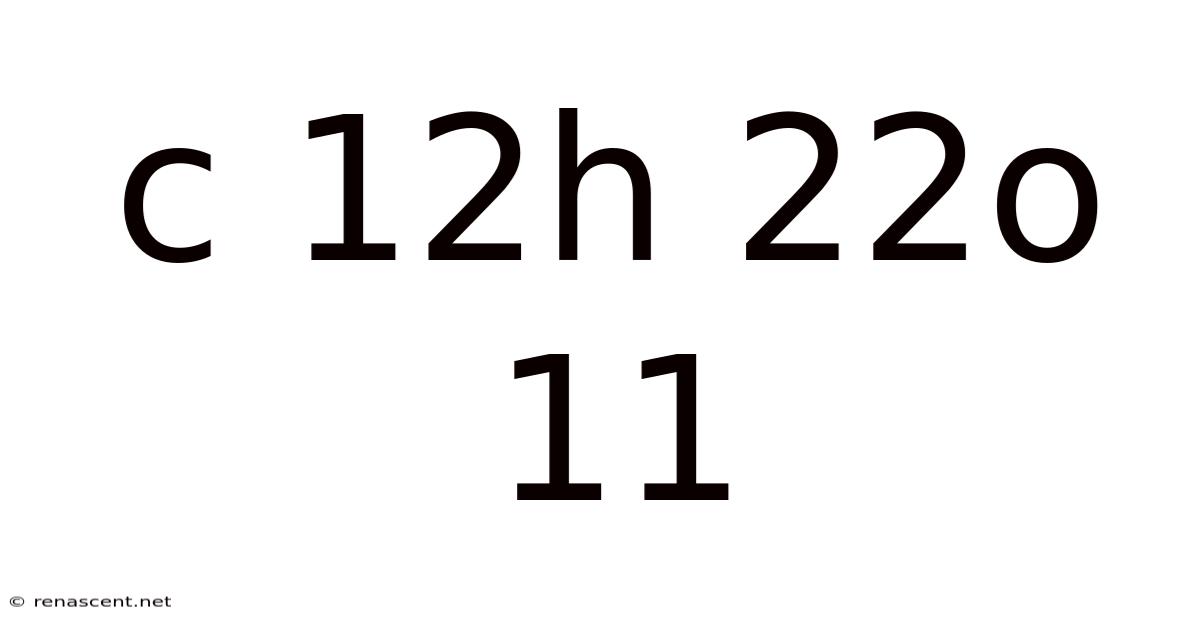Decoding the Sweetness: A Comprehensive Look at C<sub>12</sub>H<sub>22</sub>O<sub>11</sub> (Sucrose)
Sucrose, with its chemical formula C<sub>12</sub>H<sub>22</sub>O<sub>11</sub>, is a ubiquitous compound found in our daily lives. More commonly known as table sugar, it's a key player in the world of carbohydrates, impacting everything from our diets to industrial processes. This article delves deep into the fascinating world of sucrose, exploring its structure, properties, sources, uses, and implications for human health. Understanding C<sub>12</sub>H<sub>22</sub>O<sub>11</sub> goes beyond simply knowing its chemical formula; it’s about grasping its profound role in our world Easy to understand, harder to ignore. Still holds up..
Quick note before moving on.
Introduction: What is Sucrose?
Sucrose, a disaccharide, is a type of sugar composed of two simpler sugars: glucose and fructose. In real terms, these two monosaccharides are linked together through a glycosidic bond, specifically an α-1,β-2-glycosidic bond. That's why this specific bonding arrangement is crucial for its properties and how our bodies metabolize it. While seemingly simple in its chemical makeup, sucrose exhibits remarkable properties that make it indispensable across various fields. This article aims to provide a comprehensive overview of this essential carbohydrate, covering its chemical structure, physical properties, biological functions, industrial applications, and health considerations.
The Chemical Structure of C<sub>12</sub>H<sub>22</sub>O<sub>11</sub>
The chemical formula C<sub>12</sub>H<sub>22</sub>O<sub>11</sub> represents the empirical formula of sucrose. On the flip side, to fully understand its properties, we must dig into its structural formula. Sucrose is a non-reducing sugar, meaning it doesn't readily react with oxidizing agents like Benedict's solution. This is because the anomeric carbons of both glucose and fructose are involved in the glycosidic bond, preventing them from acting as reducing agents.
The molecule's structure can be visualized as a glucose molecule linked to a fructose molecule. Also, the glucose component exists in its pyranose form (a six-membered ring), while the fructose component is in its furanose form (a five-membered ring). This specific linkage is crucial for determining the molecule's overall shape, sweetness, and reactivity. Now, the α-1,β-2-glycosidic bond connects the carbon atom at position 1 of the α-D-glucopyranose unit to the carbon atom at position 2 of the β-D-fructofuranose unit. Understanding this layered structure is fundamental to comprehending its interactions with enzymes and its subsequent metabolic pathways Worth knowing..
Physical Properties of Sucrose
Sucrose is a white, crystalline solid at room temperature. It's readily soluble in water, a property exploited extensively in its various applications. Its solubility varies with temperature, increasing significantly as temperature increases. This characteristic is important in processes involving crystallization and purification of sucrose. Sucrose has a high melting point, typically around 186°C (367°F), above which it undergoes caramelization, a process involving decomposition and the formation of various brown-colored compounds, giving caramel its characteristic flavor and color. Sucrose also possesses a high refractive index, contributing to its visual appeal in crystalline form. Its sweetness is a key characteristic, a property used to enhance the taste of various food and beverages.
Sources of Sucrose
The primary source of sucrose is sugarcane ( Saccharum officinarum) and sugar beet (Beta vulgaris). That's why sugarcane, a tropical grass, stores sucrose in its stalks. On the flip side, sugar beet, a root vegetable, accumulates sucrose in its roots. The extraction process involves crushing the sugarcane or beets, followed by purification steps to obtain highly refined sucrose crystals. Both are cultivated on a massive scale globally to meet the world's insatiable demand for sugar. The process is highly energy-intensive, highlighting the significant environmental footprint associated with sugar production. Other sources of sucrose, albeit in much smaller quantities, include various fruits and vegetables, contributing to their natural sweetness.
Industrial Uses of Sucrose
Sucrose's versatility extends far beyond its role as a sweetener. Its numerous industrial applications include:
-
Food Industry: This is by far the largest application of sucrose. It's used as a sweetener in countless food products, ranging from baked goods and confectionery to beverages and processed foods. Its role extends beyond sweetness, influencing texture and preservation in various food items But it adds up..
-
Pharmaceutical Industry: Sucrose is employed as a filler and excipient in the formulation of tablets and other pharmaceutical products. It serves as a sweetener in syrups and other liquid medications, improving palatability. Its properties help with the even distribution of active ingredients and enhance the stability of the final product Took long enough..
-
Chemical Industry: Sucrose undergoes chemical modifications to yield various useful chemicals, such as ethanol through fermentation and other derivatives through chemical reactions. These derivatives find applications in diverse areas, including the production of plastics and detergents Not complicated — just consistent..
-
Biofuel Production: Sucrose is a valuable feedstock for the production of bioethanol, a renewable fuel source. This process leverages the fermentation capabilities of microorganisms to convert sucrose into ethanol, a sustainable alternative to fossil fuels.
Metabolism of Sucrose: Digestion and Absorption
When we consume sucrose, it undergoes digestion in the small intestine. Practically speaking, the enzyme sucrase, present in the brush border of the intestinal cells, hydrolyzes the α-1,β-2-glycosidic bond, breaking down sucrose into its constituent monosaccharides: glucose and fructose. So these monosaccharides are then absorbed into the bloodstream through specialized transport proteins. Glucose is the primary energy source for our cells, while fructose undergoes metabolism in the liver. In real terms, the efficiency of sucrose digestion and absorption depends on various factors, including the individual's health status and the presence of other components in the diet. Individuals with sucrase deficiency experience digestive issues upon consuming sucrose Easy to understand, harder to ignore..
Health Implications of Sucrose Consumption
While sucrose provides energy, excessive consumption is linked to various health problems. These include:
-
Weight Gain and Obesity: Sucrose is energy-dense but lacks essential nutrients. Excess consumption contributes to weight gain and an increased risk of obesity, a major risk factor for numerous chronic diseases That's the whole idea..
-
Type 2 Diabetes: Regular consumption of high levels of sucrose can lead to insulin resistance and an increased risk of developing type 2 diabetes.
-
Dental Caries (Cavities): Sucrose serves as a substrate for bacterial fermentation in the mouth, leading to the production of acids that erode tooth enamel, causing cavities.
-
Cardiovascular Disease: High sucrose intake is associated with increased levels of triglycerides and low-density lipoprotein (LDL) cholesterol, both contributing to cardiovascular disease risk Simple, but easy to overlook..
-
Non-alcoholic Fatty Liver Disease (NAFLD): Excessive fructose intake, a component of sucrose, is implicated in the development of NAFLD.
Moderate consumption of sucrose as part of a balanced diet is generally considered safe, but excessive intake should be avoided to minimize potential health risks.
Sucrose vs. Other Sweeteners: A Comparison
Sucrose is often compared with other sweeteners, such as high-fructose corn syrup (HFCS), artificial sweeteners, and other natural sugars. Artificial sweeteners are non-caloric and do not contribute to weight gain, but some concerns remain regarding their long-term effects. Each has its own characteristics and implications for health. While sucrose provides energy and satisfies the desire for sweetness, HFCS contains a higher proportion of fructose, potentially raising concerns about metabolic effects. Natural sugars found in fruits and vegetables are often considered healthier options due to their association with fiber and other nutrients.
Frequently Asked Questions (FAQ)
Q: Is sucrose the same as glucose?
A: No, sucrose is a disaccharide composed of glucose and fructose, while glucose is a monosaccharide.
Q: Is sucrose a reducing sugar?
A: No, sucrose is a non-reducing sugar because its anomeric carbons are involved in the glycosidic bond.
Q: What are the sources of sucrose?
A: The main sources are sugarcane and sugar beet.
Q: What are the health risks associated with high sucrose intake?
A: High sucrose intake is associated with weight gain, obesity, type 2 diabetes, dental caries, cardiovascular disease, and NAFLD That's the part that actually makes a difference..
Q: What are the alternatives to sucrose?
A: Alternatives include HFCS, artificial sweeteners, and natural sugars from fruits and vegetables Took long enough..
Q: Can sucrose be used in industrial applications?
A: Yes, it's used in various industries, including food, pharmaceutical, and chemical industries That's the whole idea..
Conclusion: The Sweet Truth about C<sub>12</sub>H<sub>22</sub>O<sub>11</sub>
Sucrose, with its seemingly simple chemical formula C<sub>12</sub>H<sub>22</sub>O<sub>11</sub>, plays a multifaceted role in our lives. That said, its impact on human health must be carefully considered. From its essential role as a sweetener to its diverse industrial applications, sucrose is a remarkable compound. Understanding the structure, properties, and metabolic pathways of sucrose is crucial for informed decision-making regarding its consumption and utilization. Worth adding: moderate consumption as part of a balanced diet is generally acceptable, but excessive intake should be avoided to mitigate potential health risks. Further research continues to unravel the complex details of sucrose's interaction with the human body and its broader environmental impact, ensuring responsible and sustainable utilization of this ubiquitous carbohydrate Not complicated — just consistent..