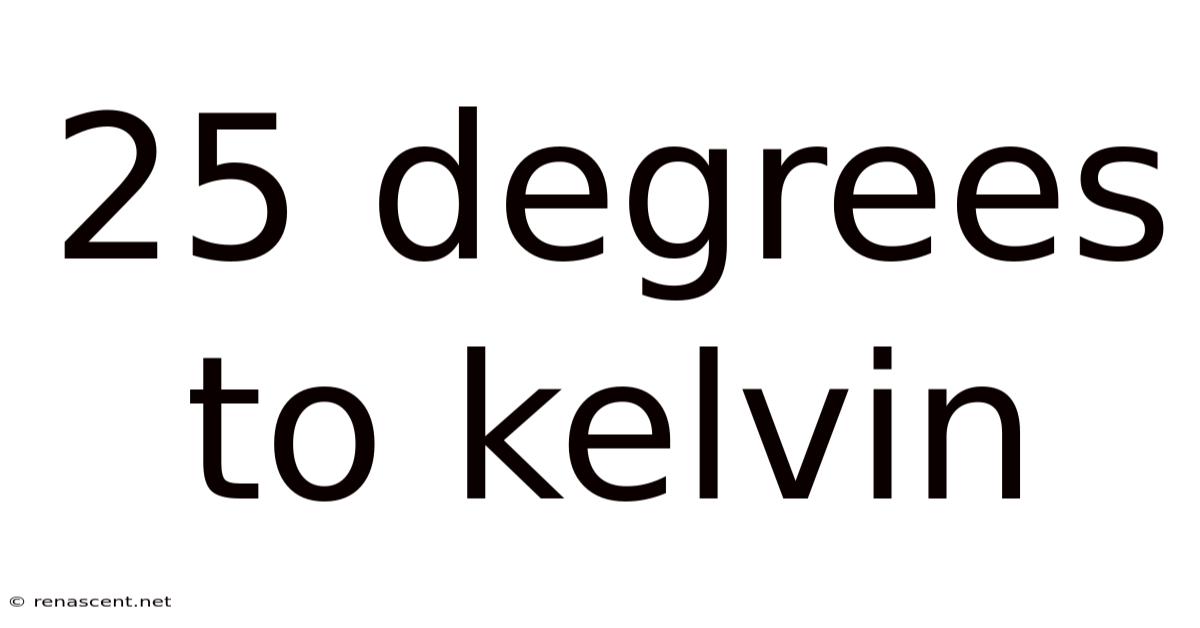Converting 25 Degrees Celsius to Kelvin: A thorough look
Understanding temperature conversions is crucial in various fields, from scientific research and engineering to everyday cooking and meteorology. This article will delve deep into the process of converting 25 degrees Celsius (°C) to Kelvin (K), explaining the underlying principles, providing step-by-step instructions, and exploring the scientific context behind this conversion. We will also address frequently asked questions and provide practical applications to solidify your understanding Still holds up..
Introduction: Celsius, Kelvin, and the Importance of Conversion
The Celsius (°C) and Kelvin (K) scales are both commonly used temperature scales, but they differ significantly in their zero points and the size of their units. Worth adding: celsius, a metric unit, uses the freezing and boiling points of water (0°C and 100°C, respectively) as reference points. That's why kelvin, on the other hand, is the base unit of thermodynamic temperature in the International System of Units (SI). Because of that, its zero point, absolute zero (0 K), represents the theoretical absence of all thermal energy. This absolute zero is equivalent to -273.15°C. Consider this: the importance of converting between these scales arises from the need for consistency and accuracy in scientific calculations and engineering applications where absolute temperature values are essential. This article focuses specifically on converting 25°C to Kelvin, a frequently encountered conversion in various contexts.
Step-by-Step Conversion: 25 Degrees Celsius to Kelvin
The conversion from Celsius to Kelvin is remarkably straightforward. It involves a simple addition:
Kelvin (K) = Celsius (°C) + 273.15
That's why, to convert 25°C to Kelvin, we simply add 273.15 to 25:
25°C + 273.15 = 298.15 K
That's why, 25 degrees Celsius is equal to 298.That said, 15 Kelvin. This simple formula holds true for any Celsius temperature you wish to convert.
A Deeper Dive into the Scientific Rationale
The relationship between Celsius and Kelvin is not arbitrary; it's rooted in the thermodynamic properties of matter. Here's the thing — the Kelvin scale is based on the concept of absolute zero, the lowest possible temperature. At absolute zero, all molecular motion theoretically ceases. This is a fundamental concept in thermodynamics and statistical mechanics But it adds up..
While Celsius uses the freezing and boiling points of water as reference points, these are merely convenient markers within a broader temperature range. The Kelvin scale, however, provides a more fundamental and absolute measure of temperature. This is particularly important in calculations involving ideal gas laws, where temperature is directly proportional to the average kinetic energy of gas molecules. Using Kelvin ensures that the calculations accurately reflect the absolute thermal energy content of the system Small thing, real impact..
Practical Applications of Celsius to Kelvin Conversion
The conversion from Celsius to Kelvin is not just a theoretical exercise; it has numerous practical applications across various disciplines:
-
Scientific Research: Many scientific experiments and calculations, especially in fields like chemistry and physics, require temperature measurements in Kelvin. Here's one way to look at it: determining reaction rates, calculating thermodynamic properties, and understanding phase transitions often necessitate Kelvin values Took long enough..
-
Engineering: In engineering, particularly in thermodynamics and fluid mechanics, Kelvin is preferred because it provides an absolute measure of temperature, crucial for accurate calculations and design considerations. This is critical in designing engines, power plants, and refrigeration systems Which is the point..
-
Meteorology: While weather reports typically use Celsius or Fahrenheit, meteorological models and climate simulations often rely on Kelvin to accurately represent atmospheric processes and predict weather patterns Worth keeping that in mind..
-
Material Science: The behavior of materials, such as their strength, conductivity, and phase transitions, is highly temperature-dependent. Using Kelvin provides a consistent and absolute measure for accurate characterization and analysis of material properties Not complicated — just consistent..
-
Astronomy: The study of stars and other celestial bodies involves extremely high temperatures, often expressed in Kelvin. Understanding the temperature of stars is crucial for determining their properties, evolution, and distance Practical, not theoretical..
-
Everyday Life (indirectly): Even in everyday situations, Kelvin plays a role indirectly. Many technological devices, from refrigerators to smartphones, operate based on principles that rely on accurate temperature measurements, often indirectly utilizing Kelvin-based calibrations.
Addressing Frequently Asked Questions (FAQs)
Q1: Why is it important to use Kelvin instead of Celsius in certain scientific calculations?
A1: Kelvin represents absolute temperature, meaning it starts at absolute zero, where all molecular motion ceases. This absolute scale is crucial for many thermodynamic calculations and gas law equations because temperature is directly proportional to the kinetic energy of molecules. Using Celsius would introduce errors and inconsistencies in these calculations.
Q2: Can I convert from Kelvin back to Celsius?
A2: Absolutely! The reverse conversion is equally straightforward:
Celsius (°C) = Kelvin (K) - 273.15
Q3: Are there any other temperature scales besides Celsius and Kelvin?
A3: Yes, the Fahrenheit (°F) scale is another commonly used scale, primarily in the United States. Converting between Celsius, Kelvin, and Fahrenheit requires more complex formulas, but the principles remain the same: aligning with the absolute temperature scale or utilizing established reference points Worth keeping that in mind..
Q4: What is the significance of absolute zero?
A4: Absolute zero (0 K) represents the theoretical lowest possible temperature, where all matter has zero thermal energy, and atomic motion ceases. While it's impossible to reach absolute zero in practice, its theoretical significance underpins many fundamental concepts in physics and thermodynamics.
Real talk — this step gets skipped all the time.
Q5: Is there a difference in the size of a degree Celsius and a Kelvin?
A5: No, the size of a degree Celsius and a Kelvin are identical. The only difference lies in their zero points Worth keeping that in mind..
Conclusion: Mastering Temperature Conversions
Understanding the conversion from Celsius to Kelvin is essential for anyone working with temperature measurements in scientific, engineering, or even everyday contexts. And the simplicity of the conversion formula, K = °C + 273. Because of that, 15, belies the profound implications of using the absolute Kelvin scale. Here's the thing — by grasping the underlying principles and practical applications, you can confidently manage temperature conversions and apply this knowledge to a wide range of situations, solidifying your understanding of fundamental scientific concepts. The ability to accurately convert between these scales is a critical skill that fosters a deeper appreciation for the interconnectedness of various scientific disciplines and real-world phenomena. Remember, a thorough understanding of the Kelvin scale and its relationship to Celsius is invaluable in numerous fields and contributes to precise scientific calculations and technological advancements.
This is the bit that actually matters in practice It's one of those things that adds up..